Paid
SERVICE DESCRIPTION | Sacrificial Anode, also known as Sacrificial Anode, is a method of protecting against corrosion of metals. The principle involves the use of two metals linked in an electrolyte, such as magnesium, zinc, and aluminum. These metals are mixed to make it…
- Laboratorium Material Serpong
- Puspiptek - Serpong
Gd. 224 Laboratorium Material Kawasan B.J Habibie Serpong, Tangerang Selatan - 08119811569
- labmaterialmetalurgi@brin.go.id
Marketing Office
Deputi Bidang Infrastruktur Riset dan Inovasi BRIN
layanan_sains@brin.go.id
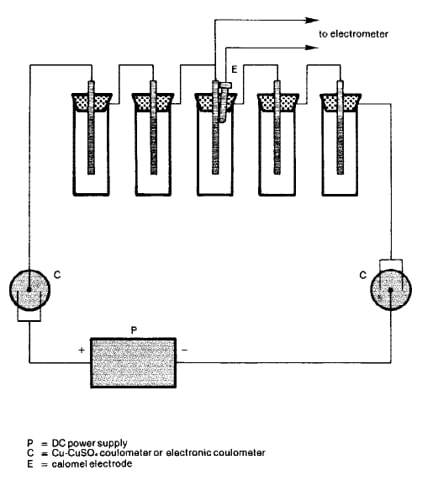
Sacrificial Anode, also known as Sacrificial Anode, is a method of protecting against corrosion of metals. The principle involves the use of two metals linked in an electrolyte, such as magnesium, zinc, and aluminum. These metals are mixed to make it more effective.
The metal that corrodes more easily (anode) discharges a protective current to protect another metal (cathode). It is important that the anode potential must be more negative than that of the structure being protected. As the structure corrodes, the protection current adapts and decreases over time.
Zinc and aluminum metals initially produce high protection currents, but will decrease over time. Therefore, it is important to routinely monitor the potential of a structure to ensure effective protection. In industry, an understanding of sacrificial anodes helps preserve the life and quality of metallic materials from corrosion.
Application Requirements:
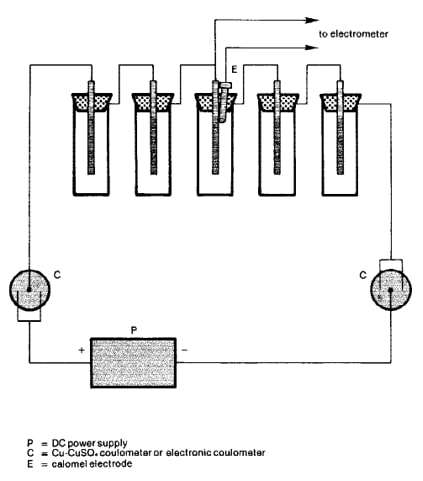
The metal that corrodes more easily (anode) discharges a protective current to protect another metal (cathode). It is important that the anode potential must be more negative than that of the structure being protected. As the structure corrodes, the protection current adapts and decreases over time.
Zinc and aluminum metals initially produce high protection currents, but will decrease over time. Therefore, it is important to routinely monitor the potential of a structure to ensure effective protection. In industry, an understanding of sacrificial anodes helps preserve the life and quality of metallic materials from corrosion.
Application Requirements:
- Photo
- Other Supporting File